In the previous section 4.25, we saw the structure of CH4 and C2H6. We also discussed sigma bond. In this section, we will see the structure of C2H4 (ethene). We will also see the basic details about pi bonds
• Fig.4.144(a) below shows the Lewis dot structure of C2H4
• The details about the model of C2H4 can be written in 13 steps:
1. Both the C atoms in C2H4 is sp2 hybridized
• Let us see how this sp2 hybridization is achieved. It can be written in 4 steps:
(i) Fig.4.144(b) below shows the orbitals in the valence shell of C
(ii) When enough energy is given, one electron in the 2s orbital jumps to the 2pz orbital
• Thus we get four half filled orbitals. This is shown in fig.c
(iii) The 2s orbital mixes together with 2px and 2py. This is shown in fig.d
(Note that, 2pz does not participate in the mixing)
(iv) Since there is one s-orbital and two p-orbitals, it is a sp2 hybridization
2. We know that:
♦ In sp2 hybridization, there will be three hybrid orbitals
✰ Together, they form a triangular planar shape
✰ The larger lobes will be directed towards the corners of a triangle
• This is shown in fig.4.145(a) below:
• Let us write four 'important points to remember' about fig.a:
(i) The three sp2 hybrid orbitals are at an angular distance of 120o apart
(ii) The nucleus of the C atom is shown as a small red sphere
♦ This sphere is situated at the origin of the coordinate axes
(iii) One of the orbitals lies exactly along the x-axis (red axis)
(iv) We know that, all the three sp2 hybrid orbitals will lie on a plane
♦ So in fig.a, all the three orbitals are lying on the xy-plane
3. Remember that, the 2pz orbital did not take part in the hybridization
♦ So it will 'remain as such' even after hybridization
♦ We have to show it in fig.a
• The modified model is shown in fig.b
• This model in fig.b represents the 'sp2 hybridized C atom'
• So we can write:
♦ The 'sp2 hybridized C atom' consists of three items:
✰ The three hybrid orbitals
✰ The 2pz orbital
✰ The nucleus (small red sphere)
• In the figs. a and b, the 'smaller lobes of hybrid orbitals' are not shown
♦ This is to reduce congestion and thus obtain greater clarity
4. Distribution of electrons in the hybrid orbitals:
• This can be written in 3 steps:
(i) We know that, the sp2 hybrid orbitals are formed from 'one s orbital' and 'two p orbitals'
(ii) In our present case of C atom, they are: 'one 2s orbital' and 'two 2p orbitals'
(iii) Before the hybridization, the orbitals mentioned in (ii) carry a total of three electrons
♦ After hybridization, the orbitals mentioned in (ii) will no longer exist
♦ Then what will happen to the three electrons?
Answer:
• The three electrons will be distributed among the three sp2 hybrid orbitals
♦ So each hybrid orbital will carry one electron
♦ This is indicated by the arrows in fig.4.145(a) above
5. Remember that, the 2pz orbital also has one electron
• So an arrow is shown in the 2pz orbital in fig.b also
6. So we have a 'sp2 hybridized C atom' in fig.4.145(b)
• We want a mirror image of that C atom
• To obtain a 'required mirror image', we need to place the mirror in the 'correct position'
• In our present case, the 'correct position' can be specified using 2 steps:
(i) The x-axis should penetrate through the mirror
(ii) The the x-axis should be perpendicular to the 'plane of the mirror'
• Such a 'correct position' is shown in fig.4.146(a) below
• The 'resulting mirror image' is also shown in the same fig.a
7. So now we have two C atoms
• Remember that, each one of them is sp2 hybridized
• We want the two C atoms to bond together
• For that we use the following four steps:
(i) We have two orbitals lying along the x-axis
♦ One belongs to the 'original C atom'
♦ The other belongs to the 'mirror image C atom'
(ii) Move the mirror image towards the original
♦ The movement should be exactly along the x-axis
(iii) When an optimum distance is reached, overlapping takes place between the 'two orbitals mentioned in (i)'
♦ This is shown in fig.4.146(b) above
• 'overlapping' means that, there will be a particular region, which will belong to both the orbitals
• This region is called 'overlapping region'
(iv) The two electrons (one from each orbital) will then lie in that 'overlapping region'
• Thus the two electrons will belong to both the C atoms
• In this way, a bond is formed between those two C atoms
• This is shown in fig.4.147(a) below:
8. So in fig.4.147(a) above, we have two C atoms which are bonded together
• Now bonding with H atoms can begin:
♦ Two H atoms will come and overlap with the free orbitals of the original C atom
♦ Another two H atoms will come and overlap with the free orbitals of the 'C atom which is the mirror image'
♦ This is shown in fig.4.147(b)
9. The structure in fig.4.147(b), is a single unit. It consists of:
♦ Two C atoms
♦ Four H atoms
• But the molecule of C2H4 is not yet formed. This is because, each of the two pz orbitals is still half occupied
10. Each of the two pz orbitals in fig.4.147(b) above needs one more electron
• So they share electrons among themselves
• Let us see how this sharing is done. It can be written in 3 steps
(i) We know that, all orbitals are electron clouds
♦ The two pz orbitals are also electron clouds
(ii) The two pz clouds overlap in a 'side-wise manner'
• The result is a cloud with 'U' shapes
♦ A 'inverted U' above the x-axis
♦ A 'upright U' below the x-axis
• This is shown in fig.4.148 below
(iii) When the overlapping is complete, the resulting cloud will contain two electrons
11. So the shape of the C2H4 molecule will be as shown in fig.4.149 below
12. Consider the plane in fig.4.150 below
♦ The two C atoms lie on that plane
♦ The four H atoms also lie on that plane
(The plane is given a bit of transparency so that, the bonding below the plane, also becomes visible)
• So the 2D representation of C2H4 will be as shown in the fig.4.150(b)
♦ It will not have any solid triangles
♦ It will not have any dashed triangles
♦ It will have only solid lines
13. Next we will see an interesting point. It can be written in 4 steps:
(i) Consider the final model in fig.4.149 above
• Let us consider it as two separate units
♦ The original C atom and it's two H atoms constitute the first unit
♦ The mirror image C atom and it's two H atoms constitute the second unit
(ii) Keep the first unit 'fixed'
♦ Rotate the second unit
♦ Nucleus of the second unit is the pivot of rotation
♦ x-axis is the axis of rotation
• This is indicated by the yellow curved arrow in fig.4.151 below
(iii) We can do this type of rotation, only by causing damage to the structure
• This is shown in fig.4.151 below:
(iv) We see that, the 'U' shaped clouds will break
♦ If they break, the two electrons in the cloud can no longer be together
♦ The bond is lost
• That is why, such a rotation is not possible
• It can be explained in 9 steps:
1. The fig.4.152(a) below shows the final structure of C2H4
2. We see that, all bonds between C and H are sigma bonds
• In other words: All C-H bonds are sigma bonds
3. We see that the C-C bond which lies along the x-axis is also a sigma bond
4. But there is a second bond between the two C atoms
• This can be explained using fig.4.152(b)
• This fig.b shows the situation before the overlapping of pz orbitals
• We can write about the 'second bond' in 4 steps
(i) Initially, the first C atom has a pz orbital
(ii) Initially, the second C atom also has a pz orbital
(iii) The above two pz orbitals overlap in a 'side-wise manner'
♦ After this overlap, the the two pz orbitals become a single orbital
♦ This is because, after the overlap, the new orbital has two electrons
(iv) When the two pz orbitals become one, 'their parent C atoms' will naturally unite
♦ Thus a second bond is formed between the two C atoms
5. Consider the two yellow lines in the fig.b
• They are the axes of the pz original orbitals
♦ Those two axes are parallel to each other
♦ Those two axes are perpendicular to the internuclear axis (the red axis)
• Even after the 'side-wise overlapping' (shown in fig.a),
♦ Those two axes remain parallel to each other
♦ Those two axes remain perpendicular to the internuclear axis (the red axis)
■ Such a bond is called pi bond
6. So we can write the definition:
• If the axes of the atomic orbitals
♦ remain parallel to each other,
♦ and also remain perpendicular to the internuclear axis,
it is called a pi bond.
♦ In symbolic form, it is written as: 𝛑 bond
7. In the Lewis structure of C2H4, we see a double bond between the two C atoms
(See fig.4.144(a) at the beginning of this section)
• A double bond is represented by putting a '=' between atoms
• So now we know that:
♦ the upper '-' in the '=' is a 𝛔 bond
♦ the lower '-' in the '=' is a 𝛑 bond
• We will see more examples of 𝛑 bond in later sections
1. The strength of a bond depends on the 'extent of overlapping' between the orbitals
♦ If there is greater overlapping, the strength will be high
♦ If there is lesser overlapping, the strength will be low
2. In 𝛔 bond, there is end-to-end overlapping
♦ So the overlapping can take place to a greater extent
3. In 𝛑 bond, the overlapping is side-wise
♦ So the overlapping can take place only to a lower extent
4. So the 𝛔 bond will be stronger than the 𝛑 bond
• Fig.4.144(a) below shows the Lewis dot structure of C2H4
• The details about the model of C2H4 can be written in 13 steps:
1. Both the C atoms in C2H4 is sp2 hybridized
• Let us see how this sp2 hybridization is achieved. It can be written in 4 steps:
(i) Fig.4.144(b) below shows the orbitals in the valence shell of C
 |
| Fig.4.144 |
• Thus we get four half filled orbitals. This is shown in fig.c
(iii) The 2s orbital mixes together with 2px and 2py. This is shown in fig.d
(Note that, 2pz does not participate in the mixing)
(iv) Since there is one s-orbital and two p-orbitals, it is a sp2 hybridization
2. We know that:
♦ In sp2 hybridization, there will be three hybrid orbitals
✰ Together, they form a triangular planar shape
✰ The larger lobes will be directed towards the corners of a triangle
• This is shown in fig.4.145(a) below:
 |
| Fig.4.145 |
(i) The three sp2 hybrid orbitals are at an angular distance of 120o apart
(ii) The nucleus of the C atom is shown as a small red sphere
♦ This sphere is situated at the origin of the coordinate axes
(iii) One of the orbitals lies exactly along the x-axis (red axis)
(iv) We know that, all the three sp2 hybrid orbitals will lie on a plane
♦ So in fig.a, all the three orbitals are lying on the xy-plane
3. Remember that, the 2pz orbital did not take part in the hybridization
♦ So it will 'remain as such' even after hybridization
♦ We have to show it in fig.a
• The modified model is shown in fig.b
• This model in fig.b represents the 'sp2 hybridized C atom'
• So we can write:
♦ The 'sp2 hybridized C atom' consists of three items:
✰ The three hybrid orbitals
✰ The 2pz orbital
✰ The nucleus (small red sphere)
• In the figs. a and b, the 'smaller lobes of hybrid orbitals' are not shown
♦ This is to reduce congestion and thus obtain greater clarity
4. Distribution of electrons in the hybrid orbitals:
• This can be written in 3 steps:
(i) We know that, the sp2 hybrid orbitals are formed from 'one s orbital' and 'two p orbitals'
(ii) In our present case of C atom, they are: 'one 2s orbital' and 'two 2p orbitals'
(iii) Before the hybridization, the orbitals mentioned in (ii) carry a total of three electrons
♦ After hybridization, the orbitals mentioned in (ii) will no longer exist
♦ Then what will happen to the three electrons?
Answer:
• The three electrons will be distributed among the three sp2 hybrid orbitals
♦ So each hybrid orbital will carry one electron
♦ This is indicated by the arrows in fig.4.145(a) above
5. Remember that, the 2pz orbital also has one electron
• So an arrow is shown in the 2pz orbital in fig.b also
6. So we have a 'sp2 hybridized C atom' in fig.4.145(b)
• We want a mirror image of that C atom
• To obtain a 'required mirror image', we need to place the mirror in the 'correct position'
• In our present case, the 'correct position' can be specified using 2 steps:
(i) The x-axis should penetrate through the mirror
(ii) The the x-axis should be perpendicular to the 'plane of the mirror'
• Such a 'correct position' is shown in fig.4.146(a) below
• The 'resulting mirror image' is also shown in the same fig.a
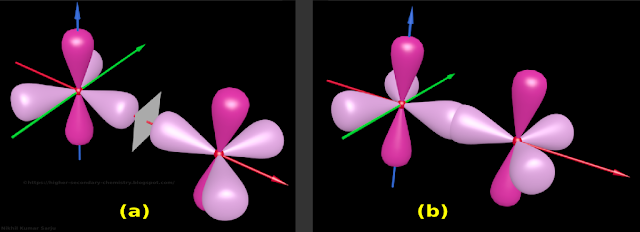 |
| Fig.4.146 |
• Remember that, each one of them is sp2 hybridized
• We want the two C atoms to bond together
• For that we use the following four steps:
(i) We have two orbitals lying along the x-axis
♦ One belongs to the 'original C atom'
♦ The other belongs to the 'mirror image C atom'
(ii) Move the mirror image towards the original
♦ The movement should be exactly along the x-axis
(iii) When an optimum distance is reached, overlapping takes place between the 'two orbitals mentioned in (i)'
♦ This is shown in fig.4.146(b) above
• 'overlapping' means that, there will be a particular region, which will belong to both the orbitals
• This region is called 'overlapping region'
(iv) The two electrons (one from each orbital) will then lie in that 'overlapping region'
• Thus the two electrons will belong to both the C atoms
• In this way, a bond is formed between those two C atoms
• This is shown in fig.4.147(a) below:
 |
| Fig.4.147 |
• Now bonding with H atoms can begin:
♦ Two H atoms will come and overlap with the free orbitals of the original C atom
♦ Another two H atoms will come and overlap with the free orbitals of the 'C atom which is the mirror image'
♦ This is shown in fig.4.147(b)
9. The structure in fig.4.147(b), is a single unit. It consists of:
♦ Two C atoms
♦ Four H atoms
• But the molecule of C2H4 is not yet formed. This is because, each of the two pz orbitals is still half occupied
10. Each of the two pz orbitals in fig.4.147(b) above needs one more electron
• So they share electrons among themselves
• Let us see how this sharing is done. It can be written in 3 steps
(i) We know that, all orbitals are electron clouds
♦ The two pz orbitals are also electron clouds
(ii) The two pz clouds overlap in a 'side-wise manner'
• The result is a cloud with 'U' shapes
♦ A 'inverted U' above the x-axis
♦ A 'upright U' below the x-axis
• This is shown in fig.4.148 below
(iii) When the overlapping is complete, the resulting cloud will contain two electrons
 |
| Fig.4.148 |
 |
| Fig.4.149 |
♦ The two C atoms lie on that plane
♦ The four H atoms also lie on that plane
(The plane is given a bit of transparency so that, the bonding below the plane, also becomes visible)
 |
| Fig.4.150 |
♦ It will not have any solid triangles
♦ It will not have any dashed triangles
♦ It will have only solid lines
13. Next we will see an interesting point. It can be written in 4 steps:
(i) Consider the final model in fig.4.149 above
• Let us consider it as two separate units
♦ The original C atom and it's two H atoms constitute the first unit
♦ The mirror image C atom and it's two H atoms constitute the second unit
(ii) Keep the first unit 'fixed'
♦ Rotate the second unit
♦ Nucleus of the second unit is the pivot of rotation
♦ x-axis is the axis of rotation
• This is indicated by the yellow curved arrow in fig.4.151 below
(iii) We can do this type of rotation, only by causing damage to the structure
• This is shown in fig.4.151 below:
 |
| Fig.4.151 |
♦ If they break, the two electrons in the cloud can no longer be together
♦ The bond is lost
• That is why, such a rotation is not possible
• The structure of C2H4 gives us an opportunity to learn about pi bond
1. The fig.4.152(a) below shows the final structure of C2H4
 |
| Fig.4.152 |
• In other words: All C-H bonds are sigma bonds
3. We see that the C-C bond which lies along the x-axis is also a sigma bond
4. But there is a second bond between the two C atoms
• This can be explained using fig.4.152(b)
• This fig.b shows the situation before the overlapping of pz orbitals
• We can write about the 'second bond' in 4 steps
(i) Initially, the first C atom has a pz orbital
(ii) Initially, the second C atom also has a pz orbital
(iii) The above two pz orbitals overlap in a 'side-wise manner'
♦ After this overlap, the the two pz orbitals become a single orbital
♦ This is because, after the overlap, the new orbital has two electrons
(iv) When the two pz orbitals become one, 'their parent C atoms' will naturally unite
♦ Thus a second bond is formed between the two C atoms
5. Consider the two yellow lines in the fig.b
• They are the axes of the pz original orbitals
♦ Those two axes are parallel to each other
♦ Those two axes are perpendicular to the internuclear axis (the red axis)
• Even after the 'side-wise overlapping' (shown in fig.a),
♦ Those two axes remain parallel to each other
♦ Those two axes remain perpendicular to the internuclear axis (the red axis)
■ Such a bond is called pi bond
6. So we can write the definition:
• If the axes of the atomic orbitals
♦ remain parallel to each other,
♦ and also remain perpendicular to the internuclear axis,
it is called a pi bond.
♦ In symbolic form, it is written as: 𝛑 bond
7. In the Lewis structure of C2H4, we see a double bond between the two C atoms
(See fig.4.144(a) at the beginning of this section)
• A double bond is represented by putting a '=' between atoms
• So now we know that:
♦ the upper '-' in the '=' is a 𝛔 bond
♦ the lower '-' in the '=' is a 𝛑 bond
• We will see more examples of 𝛑 bond in later sections
Strength of sigma and pi bonds
This can be written in 4 steps:1. The strength of a bond depends on the 'extent of overlapping' between the orbitals
♦ If there is greater overlapping, the strength will be high
♦ If there is lesser overlapping, the strength will be low
2. In 𝛔 bond, there is end-to-end overlapping
♦ So the overlapping can take place to a greater extent
3. In 𝛑 bond, the overlapping is side-wise
♦ So the overlapping can take place only to a lower extent
4. So the 𝛔 bond will be stronger than the 𝛑 bond
• In the next section, we will see the structure of C2H2 (ethyne)
No comments:
Post a Comment